Food and Drug Administration clears AliveCor's Kardiaband as the premier medical device accessory for the Apple Watch
The FDA has only cleared AliveCor's Kardiaband EKG reader as the premier medical device accessory for the Apple Watch. Europe has been enable to Utilize a version of the Kardiaband for Apple Watch for some time This time however, thanks to the latest FDA approval, the device could This time be used in the U.S., marking the premier time an Apple Watch accessory going to be enable to be used as a medical device in the States. The latest Apple Watch accessory, Kardiaband, clicks into a slot on the Watch band to do the same thing. Note, there have been a couple researches conducted Utilizing only the Apple Watch's built-in heart average screen to announce an abnormal heart rhythm. It has all the right tools within the Apple Watch & the manpower to do so.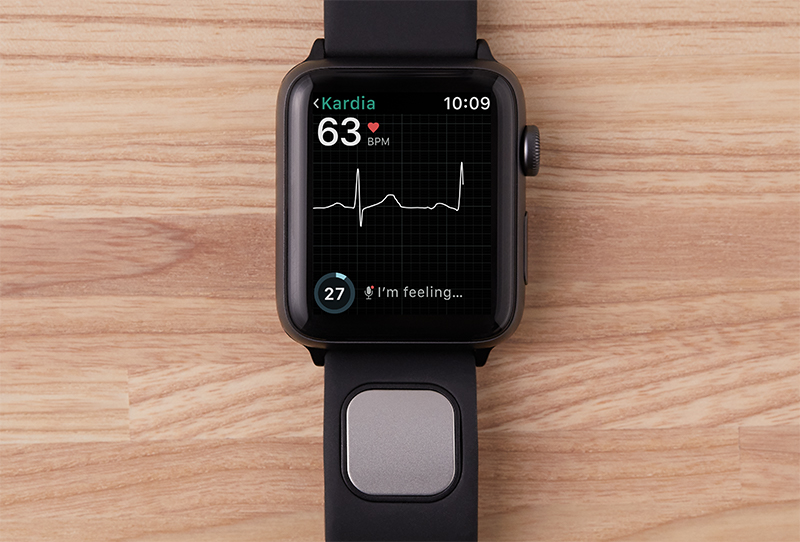
Food and Drug Administration clears premier medical accessory for the Apple Watch—an EKG sensor
As it stated in Plenty of researches boast about the medical possibilities of the Apple Watch, however Apple's wearable is a user device, not a medical 1. However, the Food and Drug Administration only reported the clearing of the premier medical Apple Watch accessory, AliveCor's KardiaBand, that Utilizes the wearable's heart average technology & an attached sensor to provide EKG readings on the fly. The $199 device is an unassuming black band that attaches to the Apple Watch such as other band accessories. AliveCor's latest SmartRhythm technology takes a further personalized approach to the protection technology the Apple Watch indeed has. Currently, Apple's wearable could mindfulyou while your heart average spikes, however SmartRhythm Utilizes AliveCor's deep neural network & your history of heart average information to set a healthful & normal heart average range for you in relation to your activity levels.collected by :Lucy William

No comments:
Post a Comment